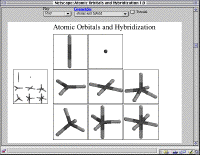
This module is a new module dealing with the concept of hybridization of atomic orbitals. The reason for it's creation is the recognition that it is often hard to 'see' how a hybrid orbital is oriented inside the 'surface' of all orbitals together.
The use of hybridizations for 3 atoms, C, Si, and Fe, allows one to compare orbital sizes between the different atoms.
This module provides the possibility to show a more in depth results of generating symmetry adapted linear combinations of atomic orbitals for different geometries, beyond the standard listed linear, trigonal planar, and tetrahedral cases. To do that it provides all symmetry allowed combinations of available orbitals in the possible sets of hybrids, e.g., the tetrahedral geometry does not only show the sp3 hybridization but also sd3 (for Si, sd3, and Fe, d3s, only).
It has the Instructor/Tutorial Versions integration and allows for Lecture Notes.
For demonstration purposes we provide the following animated gifs:
In order to provide maximum flexibility in its use at different levels of complexity, it is characterized by a minimum of words, and by a structure that allows the contents of the module to be used in a variety of orders.
The main starting point is a menu screen from which one can jump to a presentation of atomic and hybrid orbitals in C, Si, and Fe, for the geometries listed below.
The following menus are available:
The tutorial version is currently still very limited.